Neurocrine Biosciences Inc - ESG Rating & Company Profile powered by AI
Neurocrine Biosciences Inc - ESG Rating & Company Profile powered by AI
Alternative companies in the rating peer group for Neurocrine Biosciences Inc are displayed. This report of Neurocrine Biosciences Inc incorporates data from across the web and also from available disclosures by Neurocrine Biosciences Inc. Scroll down to the bottom of the page for potential risks for Neurocrine Biosciences Inc based on sector, geography and size.
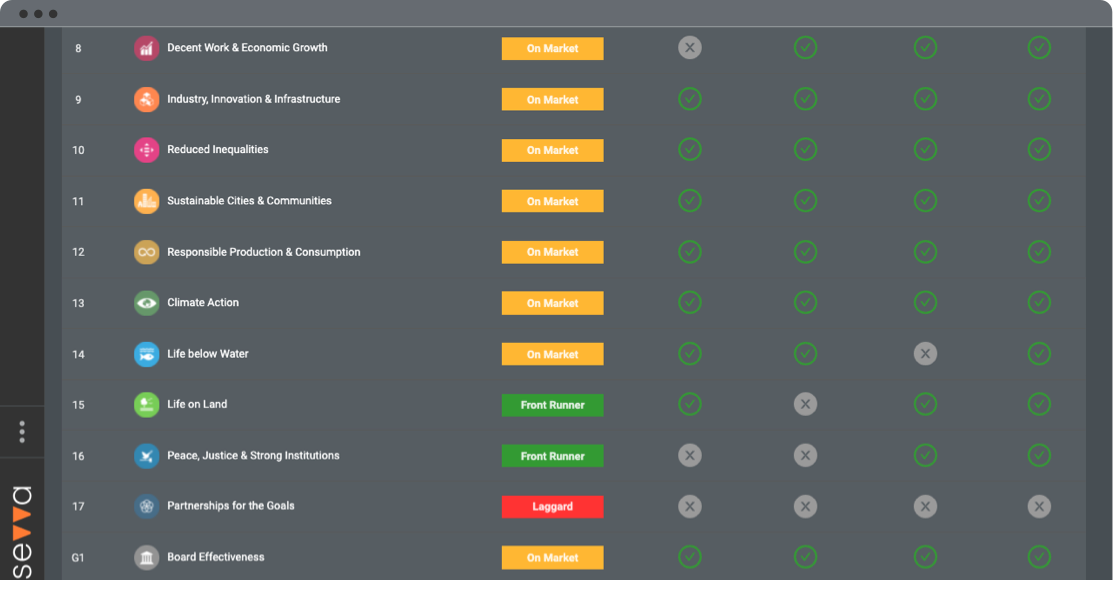
Neurocrine Biosciences Inc in the Pharmaceuticals industry gained a UN SDG ESG Transparency Score of 4.5; made up of an environmental score of 1.3, social score of 6.9 and governance score of 5.3.
4.5
Medium ImpactEnvironmental
Social
Governance
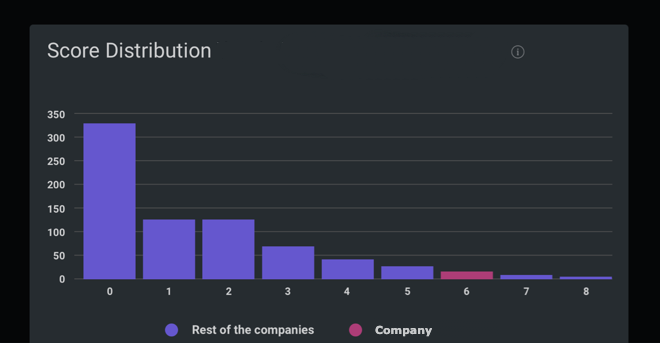
Peer Group Comparison
| Rank | Company | SDG Transparency Score ⓘ | Performance |
|---|---|---|---|
| 1 | Caladrius Biosciences Inc | 8.0 | High |
| 1 | Can Fite Biopharma Ltd | 8.0 | High |
| 852 | Vaccibody AS | 4.6 | High |
| 852 | argenx SE | 4.6 | High |
| 868 | Neurocrine Biosciences Inc | 4.5 | High |
| 868 | CARsgen Therapeutics Holdings Ltd | 4.5 | High |
| 868 | Boiron SA | 4.5 | High |
| ... | ... | ... | |
| 1970 | Zyden Gentec Ltd | 0.0 | Low |
| 1970 | iNtRON Biotechnology Inc | 0.0 | Low |
| ... | ... | ... |
Frequently Asked Questions
Does Neurocrine Biosciences Inc have an accelerator or VC vehicle to help deliver innovation?
Does Neurocrine Biosciences Inc disclose current and historical energy intensity?
Does Neurocrine Biosciences Inc report the average age of the workforce?
Does Neurocrine Biosciences Inc reference operational or capital allocation in relation to climate change?
Does Neurocrine Biosciences Inc disclose its ethnicity pay gap?
Does Neurocrine Biosciences Inc disclose cybersecurity risks?
Does Neurocrine Biosciences Inc use carbon offsets or credits exclusively for residual emissions (typically less than ~0.5–5% of total emissions)?
Does Neurocrine Biosciences Inc offer flexible work?
Does Neurocrine Biosciences Inc have a long term incentive (LTI) executive compensation plan based on a measure of return on capital?
Does Neurocrine Biosciences Inc disclose the number of employees in R&D functions?
Does Neurocrine Biosciences Inc plan to change its portfolio composition to lower the emissions intensity of its energy mix (e.g., by shifting from oil to gas, or by adding lower-carbon options like hydrogen, e-fuels, bioenergy, etc.)?
Does Neurocrine Biosciences Inc conduct supply chain audits?
Does Neurocrine Biosciences Inc disclose incidents of non-compliance in relation to the health and safety impacts of products and services?
Is there a statment that there is no plan to expand their cement production? (for example: 'We have no current plans to add additional cement making capacity')
Does Neurocrine Biosciences Inc conduct 360 degree staff reviews?
Does Neurocrine Biosciences Inc disclose the individual responsible for D&I?
Does Neurocrine Biosciences Inc disclose current and historical air emissions?
Is there a statment that there is no plan to expand their coal usage? (for example: 'We have no current plans to add additional coal powered electricity generation')
Is executive remuneration linked to climate performance?
Does the Board describe its role in the oversight of climate-related risks and opportunities?
Does Neurocrine Biosciences Inc disclose current and / or historical scope 2 emissions?
Does Neurocrine Biosciences Inc disclose water use targets?
Does Neurocrine Biosciences Inc have careers partnerships with academic institutions?
Did Neurocrine Biosciences Inc have a product recall in the last two years?
Does Neurocrine Biosciences Inc disclose incidents of discrimination?
Does Neurocrine Biosciences Inc allow for Work Councils/Collective Agreements to be formed?
Has Neurocrine Biosciences Inc issued a profit warning in the past 24 months?
Does Neurocrine Biosciences Inc disclose parental leave metrics?
Does Neurocrine Biosciences Inc disclose climate scenario or pathway analysis?
Does Neurocrine Biosciences Inc disclose current and / or historical scope 1 emissions?
Does Neurocrine Biosciences Inc explicitly state that carbon offsets or credits are separate from its emissions-reduction progress or that they are not counted toward its emissions-reduction targets?
Are Operating Expesnses linked to emissions reduction?
Does Neurocrine Biosciences Inc disclose the pay ratio of women to men?
Does Neurocrine Biosciences Inc support suppliers with sustainability related research and development?
Does Neurocrine Biosciences Inc disclose the number of operations that have been subject to human rights reviews or human rights impact assessments?
Does Neurocrine Biosciences Inc reflect climate-related risks in its financial statements?
Is there a statment that there is no plan to expand their carbon intensite energy assets? (for example: 'We have no current plans to carry out further drilling for oil,')
Is Neurocrine Biosciences Inc involved in embryonic stem cell research?
Does Neurocrine Biosciences Inc disclose GHG and Air Emissions intensity?
Does Neurocrine Biosciences Inc disclose its waste policy?
Does Neurocrine Biosciences Inc report according to TCFD requirements?
Does Neurocrine Biosciences Inc plan to mitigate emissions from future new production assets through measures such as electrifying equipment, carbon capture and storage, repurposing waste gas, methane leak detection and repair, eliminating flaring, etc.?
Does Neurocrine Biosciences Inc disclose its policies for bribery, corruption, whistle-blower, conflict of interest?
Does Neurocrine Biosciences Inc disclose energy use targets?
Does Neurocrine Biosciences Inc disclose its Renewable Energy targets?
 Subscription required
Subscription requiredAre emissions metrics verified by STBi?
 Subscription required
Subscription requiredDoes Neurocrine Biosciences Inc have a policy relating to cyber security?
Have a different question?
Potential Risks for Neurocrine Biosciences Inc
These potential risks are based on the size, segment and geographies of the company.
Neurocrine Biosciences, Inc. discovers, develops, and markets pharmaceuticals for neurological, endocrine, and psychiatric disorders. The company's portfolio includes treatments for tardive dyskinesia, Parkinson's disease, endometriosis, and uterine fibroids, as well as clinical programs in various therapeutic areas. Its lead asset is INGREZZA, a VMAT2 inhibitor for the treatment of tardive dyskinesia. The company's commercial products include ONGENTYS, a catechol-O-methyltransferase inhibitor used as an adjunct therapy to levodopa/DOPA decarboxylase inhibitors for patients with Parkinson's disease; ORILISSA for the management of moderate to severe endometriosis pain in women; and ORIAHNN, a non-surgical oral medication option for the management of heavy menstrual bleeding associated with uterine fibroids in pre-menopausal women. Its product candidates in clinical development include NBI-921352 for treating pediatric patients, as well as adult focal epilepsy indications; and NBI-827104 to treat rare pediatric epilepsy and essential tremor. The company's products in clinical development also comprise NBI-1065845 for the treatment of major depressive disorder; NBI-1065846 for treating anhedonia in major depressive disorder; and NBI-118568 for the treatment of schizophrenia. It has license and collaboration agreements with Heptares Therapeutics Limited; Takeda Pharmaceutical Company Limited; Idorsia Pharmaceuticals Ltd; Xenon Pharmaceuticals Inc.; Voyager Therapeutics, Inc.; BIAL " Portela & Ca, S.A.; Mitsubishi Tanabe Pharma Corporation; and AbbVie Inc. Neurocrine Biosciences, Inc. was incorporated in 1992 and is headquartered in San Diego, California.